National
Petrochemical & Refiners Association
1899
L Street, NW, Suite 1000
Washington, DC
20036
LW-01-137
A
US MARKET SPACE ANALYSIS OF GTL LUBRICANTS
By
Thomas F. Glenn
President
Petroleum
Trends International, Inc.
Metuchen,
NJ
Presented
at the
NPRA
Lubricants & Waxes Meeting
November 8-9, 2001
Omni Houston Hotel
Houston,
TX

Publication
of this paper does not signify that the contents
necessarily reflect the opinions of the NPRA, its
officers, directors, members, or staff.
NPRA claims no copyright in this work.
Requests for authorization to quote or use the
contents should be addressed directly to the author(s)
ABSTRACT
Gas-to-liquid
(GTL) processing provides a means to convert natural gas
to such products as diesel fuel, jet fuel, naphtha, base
oil, wax, olefins, and alcohols. The products produced by
GTL are typically exceedingly clean. In addition, they
have very favorable manufacturing economics.
Lubricant
base oils produced by GTL processing are expected in the
market in the 2005/2006 timeframe. They are expected to
initially enter the market by competing with Group III and
IV in the synthetic and synthetic-blend engine oil market
space. To a lesser extent, GTL base oils will also compete
with Group II+ as a correction fluid for Group I in 10W-30
formulations, and as a workhorse against Group II+ in a
growing market for 5W-30. Rather than reducing the value
of GTL by moving down the quality continuum to Group I and
II base oils, GTL base oils are expected to reside in the
high-end market space of Group II+, III, and IV and be the
beneficiaries of demand being pushed into this market
space by more stringent specifications.
In
addition to GTL competing in the emerging low viscosity
passenger car motor oils markets, it will also penetrate
automotive driveline applications, premium diesel engine
oils, and high-end industrial lubricant applications over
the next five to eight years.
ACKNOWLEDGEMENTS
This
paper is based on primary research conducted by
PetroTrends professional staff over the last three months.
In addition, it includes information derived from such
secondary resources as the Internet, and other public
domain documentation. It is also supplemented by
information and insights provided by Nexant Chem Systems.
Nexant Chem Systems is a market research and consulting
firm. The firm recently completed a multiclient study
focusing on the manufacturing economics of GTL; Stranded Gas Utilization: Methane Refineries of the Future."
PetroTrends
would also like to acknowledge Syntroleum for sharing its
insights on some of the typical performance
characteristics for GTL base oils.
BACKGROUND
The
technology of converting gas to liquids (GTL), is based on
the chemical process known as Fischer-Tropsch (F‑T)
synthesis. The products produced by GTL include naphtha,
kerosene, jet and diesel fuels. In addition, GTL plants
also produce such specialty products as lubricant base
oils, waxes, olefins, and alcohols.
Interest
in GTL has grown rapidly over the last five years for
several reasons. First, it provides a means to monetize
significantly more of the world’s natural gas reserves.
These reserves are estimated at over 14,000 TCF and hold
the potential to produce an equivalent of several hundred
billion barrels of crude oil. According to a study on GTL
by Arthur D. Little, an estimated “900 TCF of gas
reserves are potentially suitable for monetization by GTL
technology.” A significant percentage of these reserves
are located in regions where there is little to no
domestic demand or too far from export markets to have
much economic value.
Beyond
the value of generating more equivalent crude, however,
GTL provides an economically attractive means to produce
fuels and specialty products far cleaner then those
derived from traditional crude oil processing. This is
particularly important in light of the increasingly
stringent diesel fuel regulations coming into play. In the
US, for example, the United States Environmental
Protection Agency (EPA) will mandate a maximum of 15 parts
per million (ppm) sulfur level in diesel fuel in 2006.
Even more restrictive regulations are expected in
Europe. In May of this year, the European Commission
proposed phasing in a 10 ppm limit on sulfur starting in
2005. Similar requirements are also on the horizon in
Japan and other countries. These and other sulfur limits
on the horizon will be a significant challenge for
refiners to meet when one considers that the average level
of sulfur in much of the diesel produced today is roughly
300 to 350 ppm.
Diesel
fuel produced by the GTL process is exceedingly clean. It
has no detectable levels of sulfur or aromatics. It also
has significantly higher cetane numbers than its crude oil
derived counterpart. Diesel produced by the GTL process
can be used directly as ultra high quality fuel, or as a
blend component to boost the performance of lower quality
traditional diesel fuel. Similarly, GTL processing also
produces high quality (e.g. low sulfur, low aromatic
content) kerosene, jet fuel, naphtha and a number of such
specialty products as olefins, waxes, lubricant base oils,
and others.
In
addition to producing very high quality, environmentally
desirable “synthetic fuels, or synfuels” and specialty
products, GTL is also attracting a high degree of interest
because it provides a means to eliminate flaring and/or
reinjecting natural gas. Flaring is considered an
environmental issue and technology that eliminates it has
value. Although somewhat a longer-term issue, GTL also
holds promise as a fuel source for fuel cells. Fuel cells
are expected to begin penetrating the internal combustion
(IC) engine market in roughly five years. The reformers
used in automotive fuel cell applications will have an
appetite for only the cleanest fuels, and GTL fuel can
offer the desired level of purity.
Driven
by the opportunity to monitize natural gas, and the other
issues mentioned, interest in GTL has climbed over the
last few years. Currently there are 13 announced GTL
projects in the world. Taken together they have the
potential to produce an estimated 870 thousand barrels a
day (TBD). The
most active regions in terms of number of plants are Qatar
and Australia; three plants have been announced for each.
Egypt is also expected to be a hotbed of GTL production
with two announced plants with a combined capacity
estimated at 145 TBD, as shown in Table 1.
|
Table
1
ANNOUNCED
GAS-TO-LIQUID PLANTS AS OF OCTOBER 2001
|
|
Location
|
Planned
capacity (TBD)
|
|
Qatar
|
290
|
|
Egypt
|
145
|
|
Australia
|
115
|
|
Argentina
|
75
|
|
Trinidad
|
75
|
|
Indonesia
|
70
|
|
Iran
|
70
|
|
Nigeria
|
30
|
|
Total
|
870
|
Although
much of the current interest in GTL is tied to monitizing
stranded gas to produce high quality diesel fuel, it has
also garnered interest due to its ability to generate high
quality specialty products, including lubricant base oil,
waxes, and olefins. In fact, there are two companies
currently using Fischer-Tropsch reactions to produce
‘synthetic” waxes.
Schümann Sasol operates a plant in South Africa
and Shell operates a plant in Bintulu Malaysia. The Shell
plant uses the Fischer-Tropsch reaction in the Shell
Middle Distillate Synthesis (SMDS) process to convert
long-chain paraffinic feed into wax and other specialty
products. Both the products produced by Shell and Schümann
Sasol have very high purity and sharp hydrocarbon
distributions. These products are typically hard waxes
with very high melting points (e.g. above 200°F)
Unlike
petroleum wax, which is a mix of iso- and normal paraffins,
F-T wax is pure normal paraffin in the C20 to C60+
range. The
characteristics of F-T waxes give them a significant
advantage over traditional petroleum waxes in such
high-melt applications as hot-melt adhesives, powdered
coatings, inks, textiles, color concentrates, and
plastics. In addition, F-T waxes are also advancing into
the phase change materials (PCM) market. This includes
such applications as heating systems, food transportation,
medical devices and therapies, and other applications
where the latent heat available from phase change can be
put to work. The global market in the high melt space is
roughly 80 to 90 million pounds, valued at roughly $50
million, or about 1% of the total global wax demand.
Although F-T waxes offer clear advantages in some
applications, in others they are disadvantaged due to
normal paraffin content and narrow hydrocarbon
distribution. This hydrocarbon profile does not currently
afford the same formulation and cut point flexibility
found in petroleum waxes and in a market as diverse and
diffuse as the wax business, formulation flexibility
offers a distinct advantage to wax suppliers.
Opportunities
in the wax market and how GTL waxes might compete in this
market space do weigh into the economics of building
plants. As a result, the outlook for GTL base oil is also
a function of the outlook for wax from these plants. This
is not to say that one could not justify the economics of
a GTL base oil plant without wax, but it does suggest that
the economics of a specialty GTL plant could be improved
if high-value wax were part of the product mix. As it does
relate to the outlook for GTL base oil production,
additional background on GTL wax and how its market space
is likely to develop follows.
GTL
wax. Most
of the wax in the market today is derived from base oil
production. Although certainly a valued product,
technically it is a byproduct of classic solvent refining
– solvent dewaxing base oil production. Unfortunately,
as a byproduct of base oil production, the future of the
petroleum wax business is not in its own hands. Instead,
it is in the hands of the lube base oil unit, and times
are changing.
Lubricant
base oil manufacturers are feeling pressure to incorporate
catalytic dewaxing technology to meet increasingly
stringent base oil performance requirements. The catalytic
dewaxing process does not yield wax. Instead, the wax
molecules are cracked and isomerized into base oil, fuels,
and other fractions. The impact of this shift has been felt greatest in the North
American market. In
the last five years, a major grassroots base oil plant was
built (Excel Paralubes) using catalytic dewaxing and three
others replaced existing solvent dewaxing technology with
catalytic dewaxing. Others
are expected to follow.
In addition, Petro-Canada added ISODEWAXING
capacity to its plant in late 1996.
In addition to declines in wax supply as a result
of conversions from solvent dewaxing to catalytic dewaxing,
supply in North America has been further eroded by the
exits of several smaller base oil producers.
These exits took wax with them.
As
discussed, how the market space for GTL base oils develops
will, in part, be influenced by the business opportunities
associated with the wax market and how these opportunities
might compete with other interests. GTL projects are
considered to have the potential to greatly increase wax
supply because roughly 50% of the yield from the syngas
reactor is wax. The economics of world scale GTL plants,
however, will
be driven by demand for low sulfur diesel fuel, not wax
and other specialty products.
Beyond
the big picture economic realities of a world scale GTL
plant, a number of the major oil companies (those with the
resources to build a world scale GTL plant) would also
have to look across their businesses before heading into
the wax market. Many of the majors still produce wax from
solvent dewaxing. These companies will likely face the prospects of
cannibalizing their existing wax business should they
decide to market wax from a GTL plant. For some, this may
prove to be a losing proposition where every pound of wax
moved into the market from the GTL plant displaces a pound
of wax they have already placed in the market and produced
from its solvent dewaxing unit.
The
next likely new entrant into the F-T wax supply pool would
be a specialty products supplier with its eyes on base
oils, wax and other specialty GTL products. This would
likely be a producer with no ties to a conventional
solvent-refining/ solvent-dewaxing lube base oil plant.
Such a player would not have to consider the issue
of cannibalization and could develop the high-melt wax
market competing aggressively in an effort to grab market
share. Although a specialty products GTL player could
potentially do this, the value of this effort is
questionable since the high-melt point wax market is
fairly well balanced. It is also important to note that a
new entrant into the F-T wax market in the high-melt
market space would be competing with entrenched suppliers.
They would also be competing with PE wax suppliers. PE wax
is already a formidable competitor with F-T in the
high-melt market space.
A
new F-T wax producer could also decide to target the large
market spaces occupied by mid- and low- melts petroleum
waxes. This, however, is not a straightforward process.
F-T wax suppliers would likely find it necessary to
fractionate the wax because the C20 to C60
range of normal paraffins is too wide for most
applications. They
may also find it necessary in many applications to blend
F-T wax with petroleum waxes in order to match performance
requirements with existing expectations. Even with the
cost burden of fractionation and blending, the cost
structure for F-T wax could prove an advantage.
In assessing the magnitude of this advantage,
however, one would have to remain grounded in the fact
that a decision to compete in this market space is a
decision to compete with a large volume of byproduct
coming from lubricant base oil production.
In
summary, this means that the primary driver for GTL plants
today is high-quality, environmentally friendly diesel
fuel, not lubricant base oils, waxes, and other specialty
products. The catalysts used in a plant designed to
produce GTL fuel and the alpha value of its products do
not readily lend themselves to base oil production.
BASE OIL MARKET SPACE DEVELOPMENT
Few
question if the market for GTL base oils will develop. The
primary questions asked today are when, where and how will
it develop, and who will develop it first. In addition,
there is a good deal of interest in the economics of these
plants. Insight into these and other questions starts with
an understanding of what GTL base oils are and what level
of performance they offer.
GTL
base oils are products synthesized by a Fischer-Tropsch
reaction. These base oils have no detectable levels of
sulfur, nitrogen, or aromatics, and they are water white.
They have a very narrow hydrocarbon distribution and
excellent oxidation stability characteristics. In
addition, the lower viscosity products (e.g. less than
4cSt) are typically biodegradable. GTL
base oils with viscosity grades used in automotive engine
oil applications (4.0 to 9.0 cSt) are expected to have a
Viscosity Index in the range of 140 to 155. By comparison,
PAO has a VI of 120 to 138 for the same viscosity range.
Another
very important attribute of GTL base oils and one that
will shape its place in the market is its volatility. GTL
base oils reportedly have NOACK volatilities significantly
lower than API Group I, II/II+ and III base oils. A 4 cSt
product, for example, is reported to have a NOACK
volatility several percentage points below 10, as compared
to a typical Group III with a NOACK in the low- to mid-
teens. These performance attributes position GTL base oils
well to compete with PAO and Group II+ and III in the
automotive lubricants market space. It also suggests that
the greatest value for GTL base oils will be realized in
the automotive lubricant viscosity grade ranges of 2 cSt
to roughly 10 cSt and that alpha values for specialty GTL
product producers will likely optimize on these grades.
GTL
base oils also have excellent low temperature properties.
In fact, they appear to be only slightly
disadvantaged when compared to PAO’s cold crank
viscosities. The
pour point of GTL base oils is, however, much closer to
that of a Group II/III than it is to a PAO.
This can be addressed by the use of pour point
depressant and GTL base oils are reported to have
excellent responsiveness to methacrylate -based pour point
depressants.
In
addition to high quality, GTL base oils also have very
favorable manufacturing economics. According to a
multiclient study recently completed by Nextant Chem
Systems, the manufacturing costs for GTL delivered in the
US market are comparable with that of Group I, II, and
II+. Even more importantly, ChemSystems' analysis reveals
that the economics for GTL are more favorable than that of
high VI Group III, as shown in Figure 1.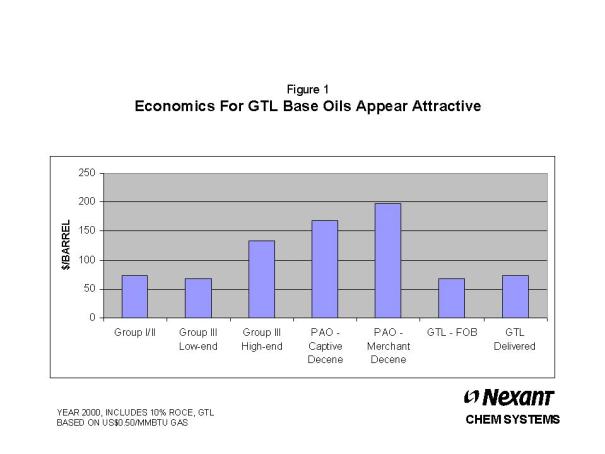
Considering
the manufacturing cost position of GTL base oils and its
performance characteristics, a starting point to begin
modeling market space development for GTL base oils is one
that looks at how the market space for API Group II and
III developed. These products also entered the market as
high performance base oils with attractive manufacturing
economics. An analysis of how the market space for Group
II and III base oils developed is provided as a backdrop
for how the market space for GTL base oils might also
develop.
Group
II and III base oils.
Group II and III base oils are product definitions that have
emerged over the last decade. The American Petroleum
Institute (API) developed the API base oil group
categories in an effort to differentiate the various
levels of base oils quality in the marketplace. In
addition to placing polyalphaolefin (PAO) in a class of
its own (GROUP IV). The system established three groups of
paraffinic base oils. These groups were based on
saturates, sulfur, and viscosity index (VI), as shown in
Table 2.
Table
2
American
Petroleum Institute Paraffinic Basestock Groups
|
|
|
Requirements
|
|
API
Group
|
Sulfur,
% wt.
|
Saturates,
% wt.
|
Viscosity
index
|
|
I
|
>
0.03 and/or
|
<
90
|
80
- 119
|
|
II
|
£
0.03 and
|
³
90
|
80
- 119
|
|
III
|
£
0.03 and
|
³
90
|
³
120
|
|
IV – a
|
-
|
-
|
-
|
|
V – b
|
-
|
-
|
-
|
|
a- Includes
polyalphaolefin (PAO).
b- Includes esters and other base oils not
included in API Groups I through IV.
|
Group
II and III base oils are generally considered superior to
Group I because they have a lower aromatic content and
higher viscosity index. Aromatic fractions tend to be more
unstable than saturated hydrocarbons, and as a result,
Group II base oils have superior thermal stability and
resistance to oxidation over Group I. In addition, as you
move up the continuum from Group II to III, you move from
base oils with a minimum VI of 95 to Group III base oils
with minimum VI over 120. This higher VI, together with
aromaticity and other issues, makes Group III base oils an
ideal blend stock to meet the more stringent volatility
requirements in passenger car motor oil. In addition, it
gives these base oils an advantage in heavy-duty motor
oil, and ATF.
Although
the API Group classifications do provide clear guidelines
to differentiate conventional and unconventional base
oils, it is important to consider the differences between
API Groups as a quality continuum based primarily on
saturates and VI, as shown in Figure 2.
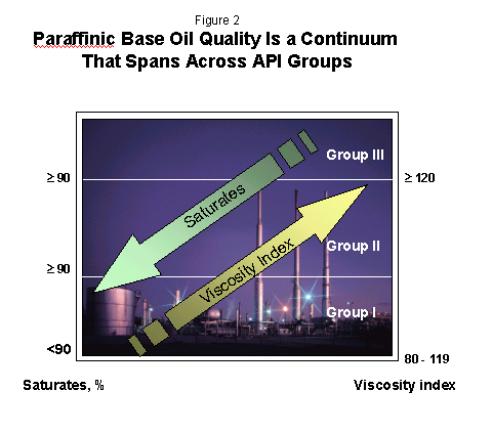
The
importance of this continuum gave rise to the “Group
II+” designation. Although Group II+ is not an official
API definition; it emerged out of the need to describe
base oils with a meaningfully higher viscosity index than
the 100 than is typical of most Group II base oils. Group
II+ base oils typically have VI in the range of 108 to
115. These base oils offer performance advantages over
Group II in some passenger car motor oil applications,
specifically related to balancing volatility with low
temperature viscometrics.
Where
GTL base oils will fit in the API base classification
system has yet to be determined. Based on some of the
performance data currently being developed, however, it is
believed that GTL base oils would likely be handled in one
of three ways. One possibility is that another API group
will be established to accommodate it. Another possibility
is that it will simply fall into a Group III designation
because it does, in fact, meet the criteria for a Group
III. Another possibility is that GTL base oils will follow
the path of Group II+. This is likely to result in a
market-place designation of Group III+.
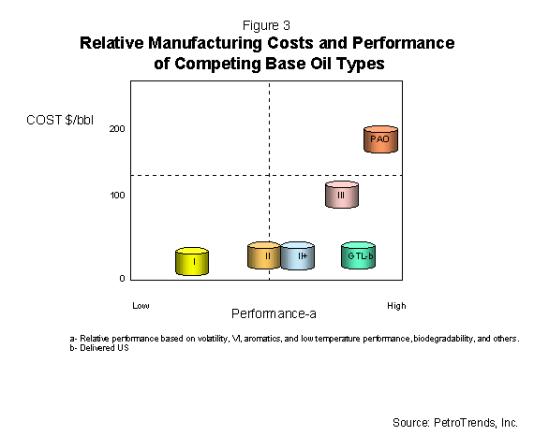
As shown in Figure 3, the performance of GTL is
considered nearly equal to Group III, however, it could
enjoy significantly lower manufacturing costs. The cost
and performance of GTL base oils suggest it will likely
track a market space development path similar to that of
III, and to a lesser extent, Group II+.
The
market space for Group II+ and Group III was developed on
several fronts, including:
- Direct
competition with PAO
- Low
volatility base oil solution for 5W-xx engine oils
- Blend
stock/correction fluid for other base oils
How the market
space for Group III and II+ developed in each of these
areas and how GTL market development might follow it is
discussed below:
Direct
competition with PAO.
Group III base oils are typically produced by incorporating
isomerization of wax fractions from the base oil into the
overall process. The isomerization process changes the
geometry of wax molecules to structures with acceptable
low temperature performance characteristics (they don’t
form wax and solidify at cold temperatures). In addition,
the isomerization of wax can significantly boost the VI of
the base oil. In fact, if run under more severe conditions
the VI of a paraffinic base oil can be pushed up to a
level that parallels that of PAO. Pushing VI up does,
however, come at the expense of yield. The high VI,
together with very low aromatic content of Group III, put
it in an excellent position to compete with PAO, and that
is exactly what it did when it entered the market.
PAO
had enjoyed a nearly unrivaled position as the
“synthetic” base oil of choice in automotive and
industrial lubricant applications.
It captured an estimated 2% of the total lubricants
market. Although PAO offered excellent oxidation stability
and unparalleled low temperature performance it had a
weakness that Group III exploited. Its weakness was
manufacturing cost. The cost to produce PAO was fairly
well studied and many were aware that the minimum costs to
produce PAO were significantly higher than that to produce
Group III. It was also well known that although Group III
could beat PAO on a cost basis, PAO still had the
virtually exclusive right to bear the valued
“synthetic” label, and PAO could far outperform Group
III in a cold crank simulator (CCS).
This advantage, however, virtually vanished
overnight when Castrol replaced PAO in its synthetic
engine oil formulation with extra high VI paraffinic base
oil. This represented a significant cost saving in the
formulation. It also resulted in a challenge from Mobil
regarding the use of the term “synthetic” by Castrol.
The challenge was brought to the National Advertising
Division (NAD) of the Council of Better Business Bureaus (CBBB).
On April 5, 1999 the NAD announced that Castrol North
America could continue to advertise its product as
“synthetic” motor oil even though Group III was being
used. Group III now had the right to wear the
“synthetic” lubricants label. Many lubricant
manufacturers switched from PAO to Group III shortly after
this ruling was announced to take advantage of the reduced
cost of the “synthetic” base oil.
In
addition to market opportunities as a replacement for PAO
in automotive applications, Group III has and will
continue to displace PAO in some industrial lubricant
applications. Its leverage in this space is, however,
weaker than it is in automotive engine oils. The
automotive engine oil segment ascribes high value to the
term “synthetic.” The industrial segment places far
less value on the term “synthetic” and much more value
on the performance advantages they offer. Although the
oxidation stability of Group III is similar to PAO, PAO
significantly outperforms Group III in low temperature
applications. As a result, market share capture by Group
III in the industrial lubricants space has come much more
slowly than in the automotive segment.
GTL
base oils have an opportunity similar to the one Group III
capitalized on in the PAO market space. The primary
difference, however, is that it will now be competing with
both PAO and Group III. Group III only had PAO to contend
with.
The
challenge for GTL in this market space, specifically in
synthetic and synthetic-blend automotive applications,
will be cost. Formulators switched from PAO to Group III
in automotive engine oils due to the cost savings one
could enjoy by blending with Group III. Any switch from
Group III to GTL would either have to represent a
relatively significant cost savings, and/or measurable
boost in performance. The performance advantages of GTL
over Group III will likely be found on several fronts. On
one front, GTL will promote the superiority of its
volatility over that of Group III. It is believed that GTL
will also use additive responsiveness and total
formulation costs as a tool to capture market share from
Group III and PAO. GTL
base oils may also provide “environmentally friendly”
solutions to the industrial lubricants market due to its
biodegradability and its absence of sulfur and aromatics.
Base
oil solution for low volatility in passenger car motor oil.
In addition to going head to head with Group III
and PAO in the high performance segment of the automotive
lubricants business, GTL is expected to compete with
Groups II+ and III with a model similar to the one used by
Group II, II+ and III to capture market share from Group I
in passenger car motor oil. It did so by responding to OEM
interests in fuel economy and the fact that the use of
lower viscosity engine oils can improve fuel economy. The
use of lower viscosity engine oils (e.g. 5W-30) did not,
however, come without concerns. In addition to the
market’s reluctance to embrace lower viscosity engine
oil grades, technical hurdles existed in regard to the
ability of the engine oil to stay in grade during use.
Engine oil can thicken and come out of grade when
subjected to the high operating temperatures in an engine
due to the light end boiling off. This meant that although
engine oil would yield desirable fuel economy performance
on an engine test stand, it did not necessarily reflect
what was actually delivered in service once the oil is
exposed to heat and aged in operation. In an effort to
address this issue, the International Lubricant
Standardization and Approval Committee (ILSAC) introduced
volatility into its GF-2 standard in the mid-1990s.
The
first iteration of GF-2 included a comparatively stringent
specification for volatility in multigrade passenger car
motor oil. It was tough, and the volatility of many of the
base oils on the market at that time did not offer the
performance necessary to meet GF-2. Base oil manufacturers
had several alternatives. One option was to narrow the
cuts in an effort to compress the hydrocarbon distribution
in the base oils. This solution was considered relatively
costly because, although it would reduce volatility by
effectively cutting off light ends, it also cut off longer
chained hydrocarbons at the other end of the distillation
curve. This approach placed a significant penalty on
yields and as a result, was costly. Another option that
could have been used to meet the first iteration of GF-2
was to blend conventional paraffinic base oil with
polyalphaolefin (PAO). This too, was considered a costly
solution because PAO was over four times the price of
conventional base oil. A third option was to work with
ILSAC and other industry stakeholders in an effort to
relax the specifications for volatility in GF-2 and give
the industry more time to prepare. The base oil industry
argued that it was not ready for such a restrictive
specification. Agreement
was reached to relax the volatility specification for GF-2
and most base oil manufacturers were then in a position to
meet the requirements.
Most
engine oils on the market at that time did come in under
the wire for the final version of GF-2. The process,
however, sent a clear message to the industry that
volatility would be revisited in the next passenger car
motor oil specification (GF-3), and that something other
than “conventional” base oil would likely be required
in the near future for those interested in competing in
the automotive lubricants business.
Although
most of the base oil in the US market was
“conventional” when GF-2 emerged, there was one
exception; Chevron. Chevron’s
Richmond plant operates with manufacturing schemes based
on hydrocraking and wax isomerization, specifically
Chevron’s ISODEWAXINGÔ
technology. Rather than removing impurities with solvents
and hydrotreating, this process uses a hydrocracking
process with special catalysts to literally break the
bonds of aromatics and saturate the remains of these and
other constituents in a high temperature, high-pressure
atmosphere that is rich in hydrogen. Unlike “conventional” solvent refining where the aromatic
content of the base oil is roughly 10%, hydrocracking
typically reduces the aromatic content of paraffnic base
oils to less than 1%. In addition, it typically produces a more refined cut in
terms of hydrocarbon distribution. These attributes, with
the catalytic dewaxing process that increases viscosity
index, resulted in base oils that could meet the more
stringent volatility requirements initially proposed in
GF-2 and beyond.
Interestingly,
although Group II base oils have been in the North
American market for close to 15 years and demonstrate
superior performance capabilities, they didn’t receive
much attention until about the last seven years. The primary reason was limited supply. As discussed, there
were only two producers in North America when GF-2 emerged
– Chevron and later Petro-Canada.
This changed, however, when Excel Paralubes (a
joint venture between Pennzoil and Conoco) built a
grassroots Group II plant that came on stream in 1997. The
Excel plant increased supply of Group II by nearly 20 TBD.
This additional supply gave Group II the critical mass
necessary to help convince automotive OEMs that the
lubricants industry now had the technology in place
required to meet more stringent specifications around
volatility. The new specification represented a step
change in PCMO volatility, as shown below in GF-3.
Table 3
NOACK Volatility
|
|
NOACK
Volatility (a)
|
|
PASSENGER
CAR MOTOR OIL GRADE
|
GF-1
|
GF-2
|
GF-3
|
|
0W‑
and 5W‑ multiviscosity grades
|
25
|
22
|
15
|
|
All
other multivisosity grades
|
20
|
22
|
15
|
|
NOTE:
(a) D-5800-99 standard test method for evaporation loss
of lubricating oils by the NOACK method.
|
This
specification would clearly favor the use of Group II and
pull through demand based on technical need. In fact, for
some grades, the specifications virtually required the use
of Group II and II+.
In addition, Group II was also showing promise as
value-added base oil in heavy-duty motor oil applications
and ATF. This too resulted in pull-through demand.
As
discussed later in this paper, GTL base oils likely will
be the beneficiaries of the momentum in pull through
demand established by Groups II, II+, and III in
automotive engine oil applications.
BLEND
STOCK/CORRECTION FLUID FOR OTHER BASE OILS
Although
base oil manufacturing is clearly shifting from Group I to
Group II in the US and Canada, Group I base oils are
expected to remain an important part of the supply pool.
These base oils are favored as the low cost workhorses for
a wide range of price sensitive industrial lubricant
applications. Some
lubricant blenders use Group I because they have captive
supply, others use it because it aligns well with their
product portfolios. In many cases, blenders heavily
reliant on Group I base oils will find it necessary to
bring in such high quality base oils as Groups II+, III,
and IV as a means to enhance the performance of the
workhorse Group I. An example of how a blender could use a
Group II+ to enhance the performance of a Group I can be
seen in a 10W-30 PCMO formulation. Although there are many
ways to meet the volatility requirements for GF-3 in a
10W-30, an economical option is to blend with roughly 70%
Group I base oil, 10% Group II+, and additives.
GTL
base oils are expected to compete with Group II+, III, and
IV as a blend stock to enhance the performance of Group I
base oils. Its ability to displace these competing stocks
is expected to be based primarily on performance and its
impact on total formulation costs.
GTL
BASE OIL MARKET SPACE DEVELOPMENT
GTL
base oils are positioned to track the footsteps already
established by Group II and II+ as the workhorse in some
multigrade engine oils and as a correction fluid in
others. The challenge for GTL base oils in the US,
however, will be the relatively sluggish market
penetration of 5W-30. In addition, Group II and II+ base
oils have already established themselves as the solution
for 5W- and 10W-30 engine oils. This means that additives
are well on their way to being optimized, blenders are
comfortable working with these stocks, and product
development costs have been invested.
Rather
than potentially giving away value by competing with
Groups I and II base oil in the 10W-30 PCMO market and
others, a more likely scenario is one that allows GTL to
maintain its value by waiting for the direction of
specifications to mature the market into the market space
currently occupied by Group III and IV, and to a lesser
extent Group II+. The direction of specification has
already moved a significant volume of base oil demand out
of the Group I space and into the Group II and II+ space
in the US market. Future
specifications will continue to push demand through the
Group II and II+ space into the space occupied by Group
III, Group IV and GTL, as shown in Figure 4.
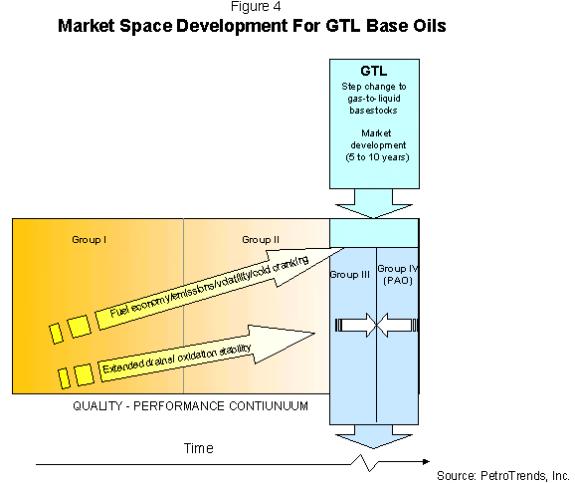
The
challenge for GTL in this approach, however, is that the
market will take time to evolve into its space. This
evolution will be tied in a large way to market acceptance
of 5W- and 0W-xx engine oils.
The most significant pull-through demand for GTL
base oils in PCMO will, however, likely be tied to 0W-xx.
Meeting the volatility requirements in this grades is
expected to be attainable only with PAO and likely GTL.
Although the low temperature performance of GTL base oils
could be an issue, data exist to suggest that this issue
can be overcome by GTL’s favorable responsiveness to
additives. It is also important to consider, however, that
even with OEMs promoting the use of 0W-xx, consumers have
the final say. If market acceptance of 5W-30 is any
indication, consumers are slow to accept lower viscosity
grades even when OEMs recommend them.
What
this means is that GTL will not likely be a significant
demand event in the US for at least the next eight to ten
years. From a product life-cycle perspective, we will
likely see GTL entering the supply pool in the 2005/2006
timeframe. If one uses the GTL plant completion schedules
currently tabled, the supply build model of Group II/II+
and III, and the grade switching rates of 5W-30 as a guide
to model with, the introduction phase of the GTL life
cycle will likely begin in 2005 and take about five years
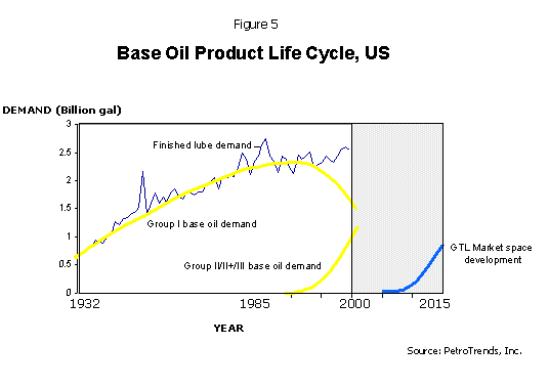
before it advances into the growth phase, as shown in Figure 5.
Initially it will do so at the expense of Group III and IV
base oils by capturing market share in the synthetic and
synthetic-blend automotive lubricant market space. It will
also penetrate the ATF and automotive driveline market
space at the same time. Market acceptance of GTL is,
however, expected to be modest during this introductory
phase of its life cycle due to a limited number of
suppliers.
GTL
is expected to transition into a growth phase by capturing
demand away from Group II, II+, III, and IV as demand for
5W- and 0W-xx PCMO increases. As additional supply comes
on line it will give OEMs and blenders the assurances they
need that supply lines are adequate and secure. This will
catalyze growth-phase demand by moving it into a
push-demand scenario similar to that currently occurring
with Group II base oils. Push marketing will drive up
demand for GTL in heavy-duty engine oil and industrial
high performance industrial applications.
GTL
is also expected to capture significant market share of
the automotive driveline segments over this same period
due to fill-for-life initiatives.
It
is also important to consider that although GTL may not be
a significant event in the US over the next eight years,
it will enjoy more aggressive growth in Europe and Asia.
The lubricants market in Europe is more mature than that
in the US and market acceptance of 5W- and 0W-xx is
further along.
CONCLUSION
Although
the primary focus of gas-to-liquid (GTL) technology is
currently on opportunities in diesel fuels, base oils
derived from this technology could also be in place by
2005. Base oils produced by GTL processing are expected to
deliver quality superior to Group III and at very
competitive costs.
Base
oils produced by GTL processing are expected to initially
enter the lubricants market by competing with Group III
and IV in the synthetic and synthetic-blend engine oil
market space. They will compete with these base oils
primarily on performance and secondarily on price and
total formulation costs. To a lesser extent, GTL base oils
will also compete with Group II+ in a growing market for
5W-30. Rather than reducing the value of GTL by moving
down the quality continuum to Group I and II base oils,
GTL base oils are expected to park themselves in the
high-end market space of Group II+, III, and IV and be the
beneficiaries of specification pushed demand into its
space. This will occur by increasingly stringent
performance requirements and market acceptance of 5W- and
most importantly 0W-xx PCMO.
In
addition to GTL competing in the emerging low viscosity
passenger car motor oils markets, it will also penetrate
automotive driveline applications, premium diesel engine
oils, high-end industrial lubricant applications, and
white oil applications over the next five to eight years.
Adoption of GTL base oils is expected to occur at a faster
rate in Europe than in the US due to the rate of market
acceptance of 0W-xx engine oils. In addition, GTL will
penetrate the Asian market.
Copyright © Petroleum
Trends International, Inc. 2002
|
![]()

![]()
![]()
